 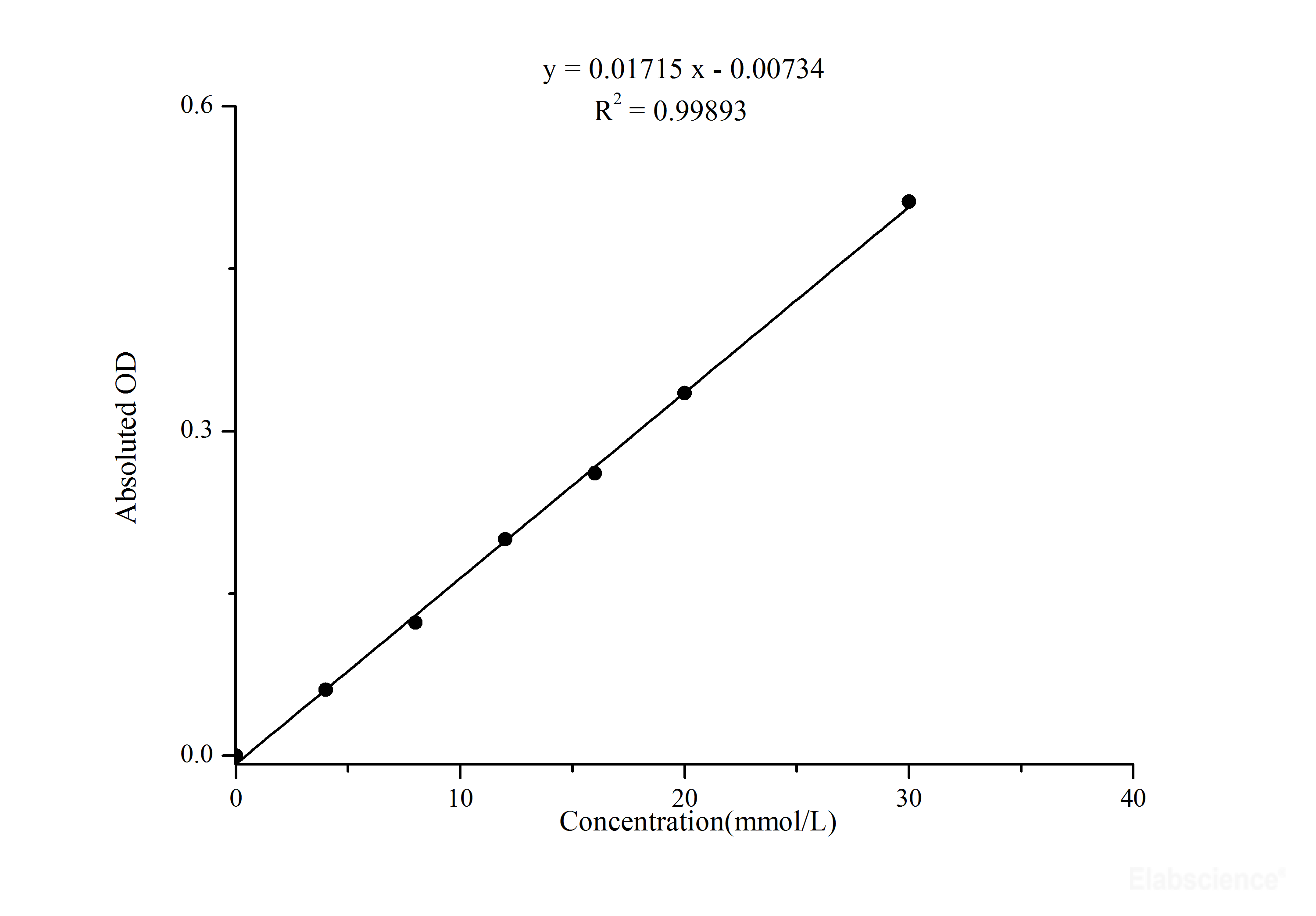 
Protein concentration is determined by reference to a standard curve consisting of known concentrations of a purified reference protein. Protein samples are added to the reagent, producing a color change or increased fluorescence in proportion to the amount added. To overcome these disadvantages, several colorimetric and fluorescent, reagent-based protein assay techniques have been developed that are used by nearly every laboratory involved in protein research. In addition, any non-protein content that absorbs UV light will interfere with measurements. UV absorption method is not ideal for protein mixtures as different proteins have differing aromatic amino acid content- varying the absorption characteristics. Direct UV-light absorbance and HPLC-reagent approaches have particular disadvantages that make these methods impractical for use with typical protein samples in proteomics workflows. Another method, traditionally used in amino acid analysis by HPLC, is to label all primary amines (i.e., N-terminus and side-chain of lysine residues) with a colored or fluorescent dye such as ninhydrin or o-phthalaldehyde (OPA). Proteins and peptides absorb UV-light in proportion to their aromatic amino acid content and total concentration. Amino acids containing aromatic side chains (i.e., tyrosine, tryptophan and phenylalanine) exhibit strong UV-light absorption. The simplest and most direct assay method for protein concentration determination in solution is to measure the absorbance at 280 nm (UV range). Depending on the accuracy required and the amount and purity of the protein available, different methods are appropriate for determining protein concentration. Protein quantitation is often necessary before processing protein samples for isolation, separation and analysis by chromatographic, electrophoretic and immunochemical techniques. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
